Vocalis Health’s COVID-19 Voice Test Achieves More than 80% Accuracy in Clinical Study
 The voice test for COVID-19 developed by Vocalis Health will accurately determine infection 81.2% of the time, according to the results of a major clinical study conducted by the Israeli startup in India last year. The test, named VocalisCheck, is being pitched as a way to augment the existing tests, saving the more traditional chemical tests for those who are at higher risk of infection.
The voice test for COVID-19 developed by Vocalis Health will accurately determine infection 81.2% of the time, according to the results of a major clinical study conducted by the Israeli startup in India last year. The test, named VocalisCheck, is being pitched as a way to augment the existing tests, saving the more traditional chemical tests for those who are at higher risk of infection.
Sound Check
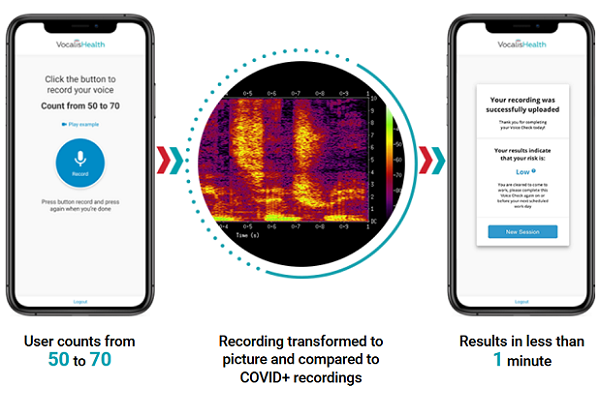
The Vocalis diagnostic test, which runs on a smartphone or computer, asks the user to count from 50 to 70. The audio is translated into a visual representation of their voice, a spectrogram, made up of 512 features vocal biomarkers. Vocalis applies artificial intelligence to compare the spectrogram to a composite image from the voice of many people proven to have COVID-19. Vocalis has been gathering public voice samples since April and started coordinating with the Israeli Ministry of Defense to get spectrograms of those who definitely had been infected.
In less than a minute, VocalisCheck would render a verdict, which would then be validated or disproven by a subsequent swab test. Vocalis worked with the Municipal Corporation of Greater Mumbai (MCGM) to run the -month pilot program of its vocal test at a medical center set up during the COVID-19 pandemic, testing more than 2,000 people speaking English, Hindi, Marathi, and Gujarati.
“The results from India are excellent for a screening tool for COVID-19,” Chief Medical Officer at Vocalis Health chief medical officer Dr. Shady Hassan told Voicebot in an interview. “But, we will bring in continuous data for [the test]. It’s important to remember VocalisCheck is not intended to replace PCR testing. It’s for screening, not diagnosing.”
“There are many use cases for the test. Big employers could do voice checks for COVID and confirm [the office] is a safer environment. Government agencies could use it to control the pandemic more effectively,” Hassan said. “The threshold of detection for the test can be adjusted to the use case too. If you’re interested it can be at high sensitivity, with the cost of some false positives. If you’re not interested in high sensitivity and false positives it can be lowered.”
The sensitivity tuning makes the test useful in more contexts. Future event organizers might want the test to be extra sensitive if they have an indoor venue with a lot of people who may have conditions that make the virus particularly dangerous. On the other hand, a small group of people who have been careful and have no reason to think they’ve been exposed might want to use the test just to be sure, but not feel the need for that level of sensitivity.
Approving Tones
Vocalis isn’t the only company developing this kind of AI-powered coronavirus diagnostic, though. Fellow Israeli tech firm Voca.ai has also been working on the idea, partnering with Carnegie Mellon University to build a database of voices for assessing COVID-19 infection. That test analyzes the probability of infection by examining the sound of someone’s voice and their cough. However, it’s not clear if Snapchat’s acquisition of Voca.ai in November for $70 million altered those plans. Indian startup Salcit Technologies built its own cough-based coronavirus test called kAs. Hassan sees these efforts as a net positive for Vocalis, rather than rivals.
“I think we as vocal biomarker companies should work together,” Hassan said. “It’s a new market that needs to be opened. We need to collaborate in order to do that.”
More data makes more accurate tests, always
Vocalis brings unique advantages to such collaborations, Hassan added. As a medical device company, it has to pass quality checks that others may not have dealt with yet. That includes a CE mark indicating Vocalis has passed all of the relevant quality and safety tests to sell VocalisCheck in the European Union. The CE mark for VocalisCheck opens the doors to a huge market, and Vocalis has ambitions that go well beyond COVID-19.
“We’re a platform and we have several stages of development focused on respiratory illness, but also on other illnesses,” Hassan said. “We have a product to detect shortness of breath in people with lung disease. We are developing tests for pulmonary hypertension, and for mental health like depression and for alcohol intoxication. We are integrating the [COVID] data with our data, after validating and standardizing, for other diseases. More data makes more accurate tests, always.”
Follow @voicebotai Follow @erichschwartz
Vocalis Health Completes COVID-19 Voice Test Pilot in Mumbai
EXCLUSIVE: A Voice Tech Startup is Gathering Data to Build a Coronavirus Speech Test